- A given atom will absorb and emit the SAME frequencies of
electromagnetic (E-M) radiation.
- A gas of hydrogen atoms will produce an absorption line spectrum
if it is between you (your telescope+spectrograph) and a continuum light
source, and an emission line spectrum if viewed from a different angle.
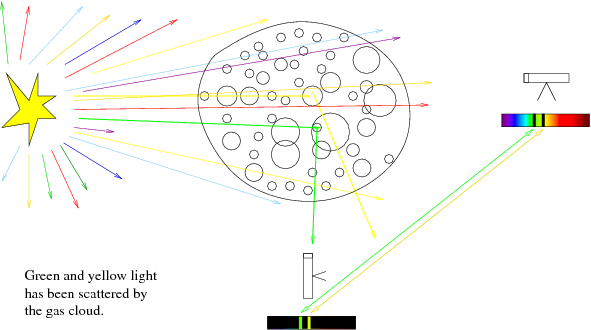
- If you were to observe the star (a source of white light) directly, you
would see a continuous spectrum, with no breaks.
- If you observe the star through the gas (telescope to right of gas
cloud, points towards star through cloud), you will see a continuous spectrum
with breaks where specific wavelengths of energy have been absorbed by the gas
cloud atoms and then re-emitted in a random direction, scattering them
out of our telescope beam. We call this an absorption spectrum (continuous +
dips).
- If you observe the gas but not the star (telescope below gas cloud,
points through gas but away from star), you will only see a portion of the
scattered light being re-emitted by the gas. The continuum radiation from the
star will not fall into our telescope beam, because we are pointed away from
the star. This is called an emission spectrum (just peaks, not continuous).
- The E-M radiation frequencies absorbed and emitted match the allowed
energy levels in the atom.
- The allowed energy levels in an atom depend mostly on the electric
field configuration. Hydrogen, with one proton in the nucleus, has a
different field configuration than does Helium with two protons – this
is why the two atoms have a different energy levels and different
characteristic absorption and emission lines.
|
| [NMSU, N. Vogt] |
- Continuum spectrum:
A gas can be collisionally excited. Imagine a hot gas. The atoms are flying
around, bumping into one another and sometimes the energy of motion during the
collision will bump an electron into a higher energy level (or completely
ionize the atom, freeing the electron completely). When this electron drops
back down to lower energy, a photon is emitted. This conversion of kinetic
energy into radiant energy cools the gas.
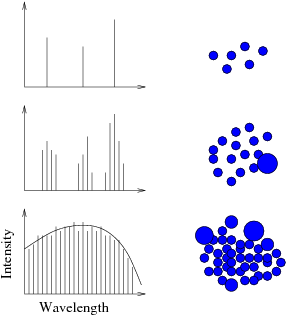
- There is a connection between emission lines from a gas and the
continuous spectrum from a solid. As you crowd atoms together (as in a
solid), the allowed energy levels in one atom start to become distorted due to
the influence of the electric field of neighboring atoms. Distort an energy
level difference a little bit and you get a slightly different frequency
emission/absorption line. A distribution of distortions leads to a
distribution of lines than eventually blend into a continuum.
- Here we see the way that a spectrum fills in as the density of
our medium increases (from gas to solid) by clumping particles together.
|
| [NMSU, N. Vogt] |
- Absorption spectrum:
What do stellar spectra look like (the light observed from stars)? Stars have
absorption line spectra. We can think of stars as a hot continuum
source with a "cool" atmosphere of absorbing gas. The wavelengths that get
absorbed depend on the chemical make up of the gas in the stellar atmosphere.
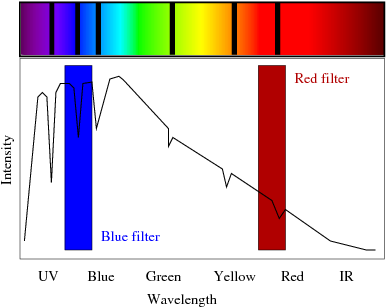
- In the 1800's the light from the Sun was dispersed and it looked
more-or-less like a Planck spectrum (a blackbody curve) with some missing
light, or absorption lines at certain wavelengths.
|
| [NMSU, N. Vogt] |
-
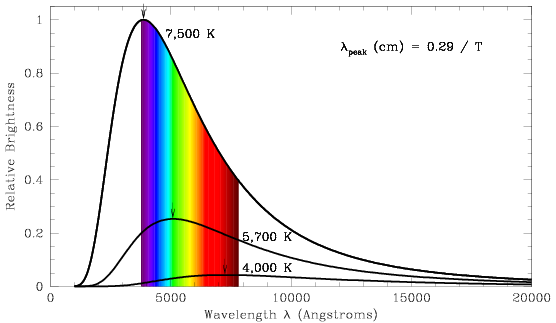
Stellar blackbody spectra have a characteristic shape, with a steep rise,
a peak in or near to the visual passband, and a slow decrease in the infrared.
Hotter stars have higher peak amplitudes, and peak at shorter wavelengths.
Blackbody curves are shown for three stars in the figure below, with
temperatures ranging from 4,000 K (a cool, red star) to 7,500 K (a hot,
violet-blue star). Small arrows mark the peak wavelength for each star.
|
| [NMSU, N. Vogt] |
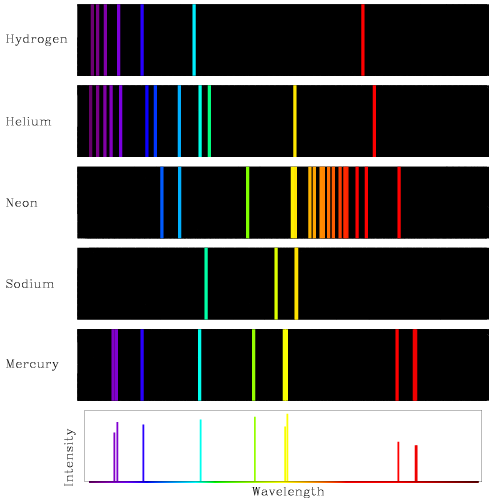
- Emission spectrum:
The wavelengths with missing light in a stellar spectrum turned out to be very
interesting and important. Their importance was realized after emission
line spectra were discovered and investigated by chemists.
- If a gas is heated to the point where it glows, the resulting spectrum
has light at discrete wavelengths that turn out to match the wavelengths of
missing light in stellar spectra. So by studying the spectra of various
elements in a laboratory here on Earth, we can determine the composition of
the distant stars!
|
| [NMSU, N. Vogt] |




